 POCl3 lewis structure consists of 3 single bonds joined to 3 chlorine atoms and a double bond with 1 oxygen atom surrounds the central phosphorous atom. One phosphorous (P), one oxygen (O) and three chlorine (Cl3) atoms in its structure. Phosphoryl oxychloride (POCl3) molecule is composed of mainly three elements i.e. POCl3 have Molecular geometry is tetrahedral. POCl3 Molecular weight is 153.33 g mol-1. POCl3 Lewis Structure How to draw lewis structure for POCl3?Ĭhemical formula for Phosphoryl oxychloride is POCl3. Lets discuss the POCl3 lewis structure in detail. 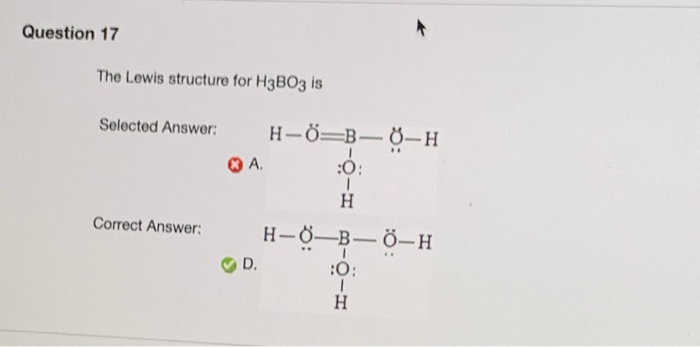
IUPAC name of phosphoryl chloride is phosphoryl trichloride. 
Phosphoryl oxychloride contains chlorine, phosphorus and oxygen atoms in its structure. The P=O bond is highly polar due to the electronegativity difference (P: 2.19, O: 3.44), influencing POCl3’s reactivity, particularly in organic synthesis as a chlorinating agent. The molecule adopts a tetrahedral geometry around phosphorus, with bond angles of approximately 109.5°. The Lewis structure displays a double bond between P and O, and three single bonds between P and each Cl atom. 
Each Cl contributes 7 valence electrons and O contributes 6, totaling 32 electrons. Phosphoryl chloride (POCl3) has a central phosphorus (P) atom with 5 valence electrons, bonded to three chlorine (Cl) atoms and one oxygen (O) atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
